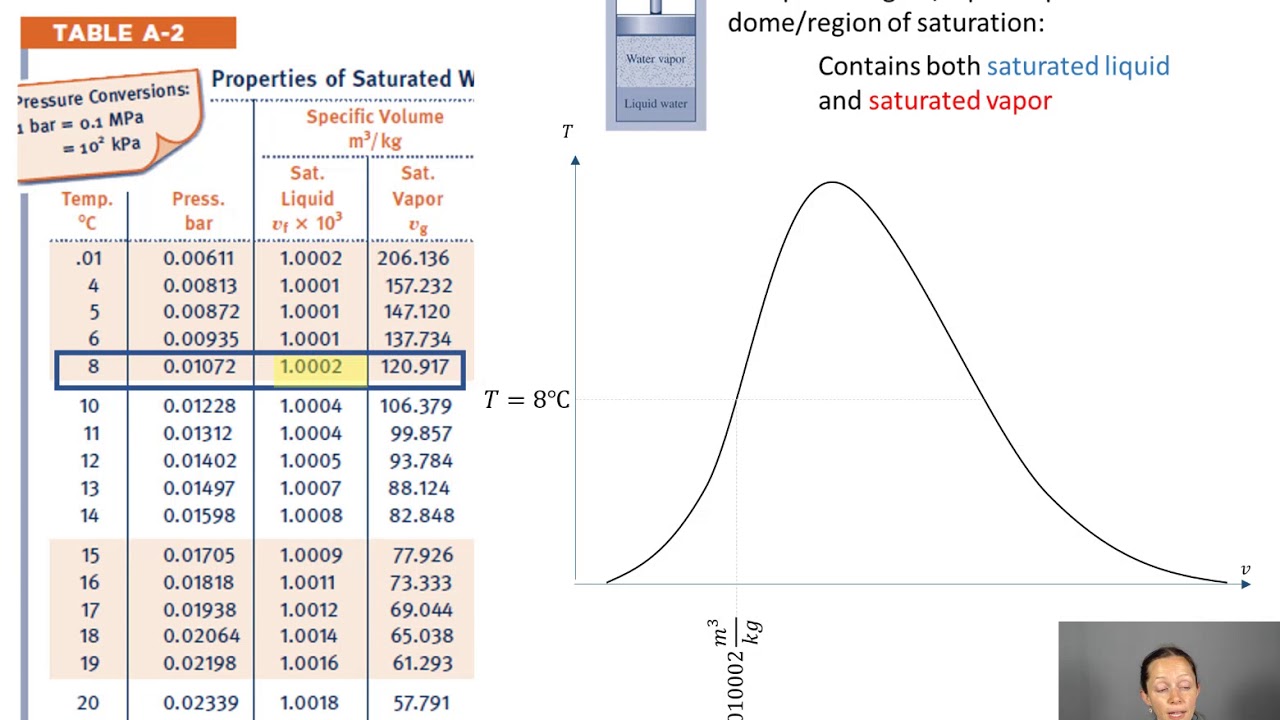
Calculate the heat needed to raise the temperature of ice until 0☌:.How much energy would you need to obtain water hot enough to brew some tea from a 1 kg block of ice with an initial temperature of -10☌ (263.15 K)? We can break it into smaller steps: The water heating calculator uses the most standard values of these constants. In this case, the units are J/kg.Īlthough sporadically considered, it's worth knowing that the value of latent heat changes with the pressure, whereas the specific heat varies depending on the temperature. 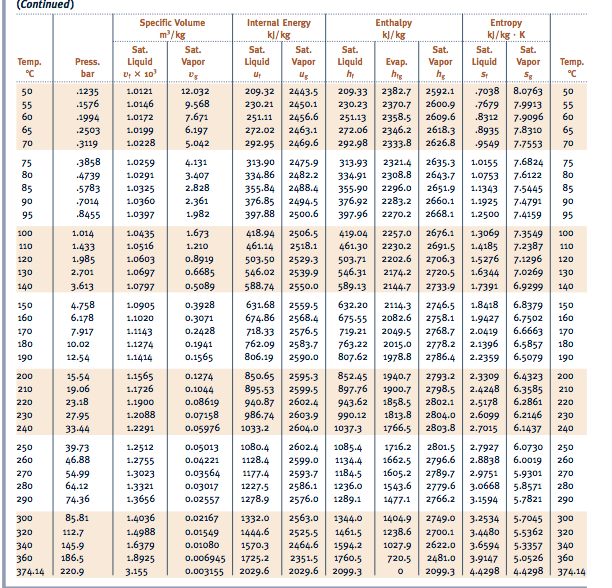
This is the amount of heat required to turn, e.g., a liquid of some mass into a gas - you could think of what happens to water at 100☌ when it becomes steam.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
